Site-Level Demo
Study-Level Demo
Patient-Level Demo
Experience Remarque systems consolidate and visualize data
 Select a document to see a common data display format
Select a document to see a common data display format
 BACK TO DOCUMENTS
BACK TO DOCUMENTS
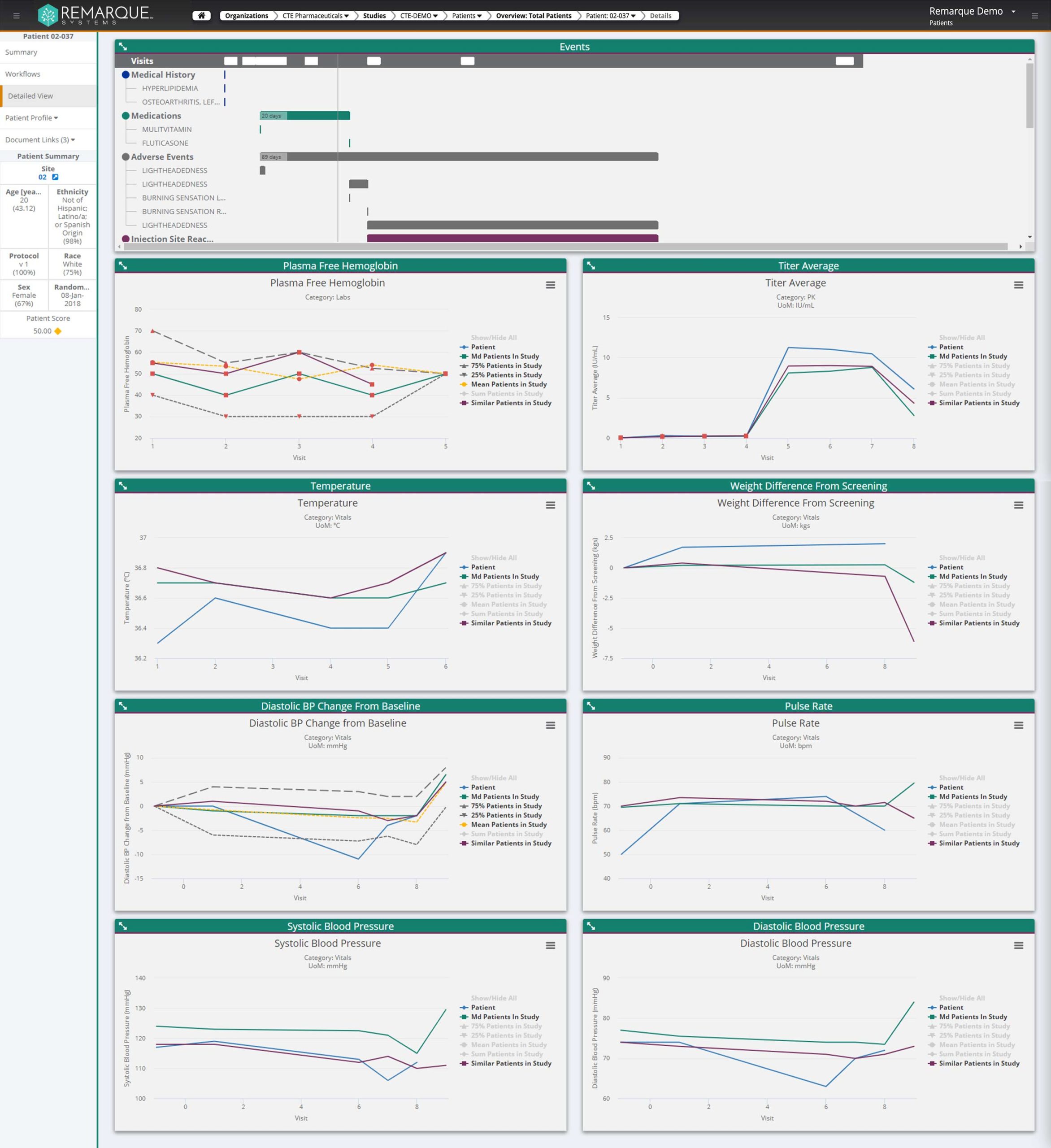
Our foundation relies on our ability to integrate all disparate data sources you may have. Our goal is to empower you to use, leverage, and gain insights from your data rather than fight and manually wrestle them together. Let us streamline this process for you–so you can focus on your area of expertise!
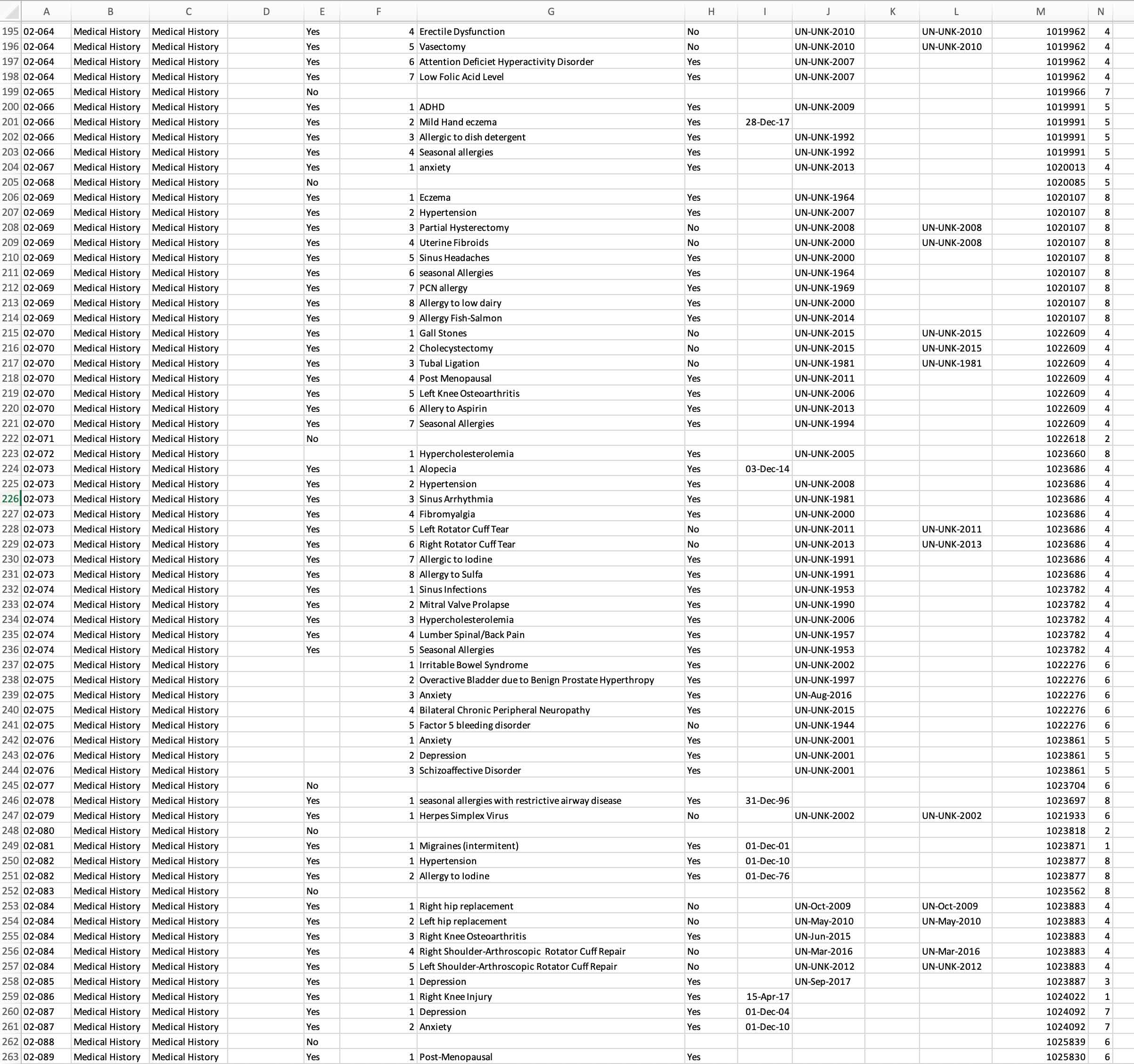
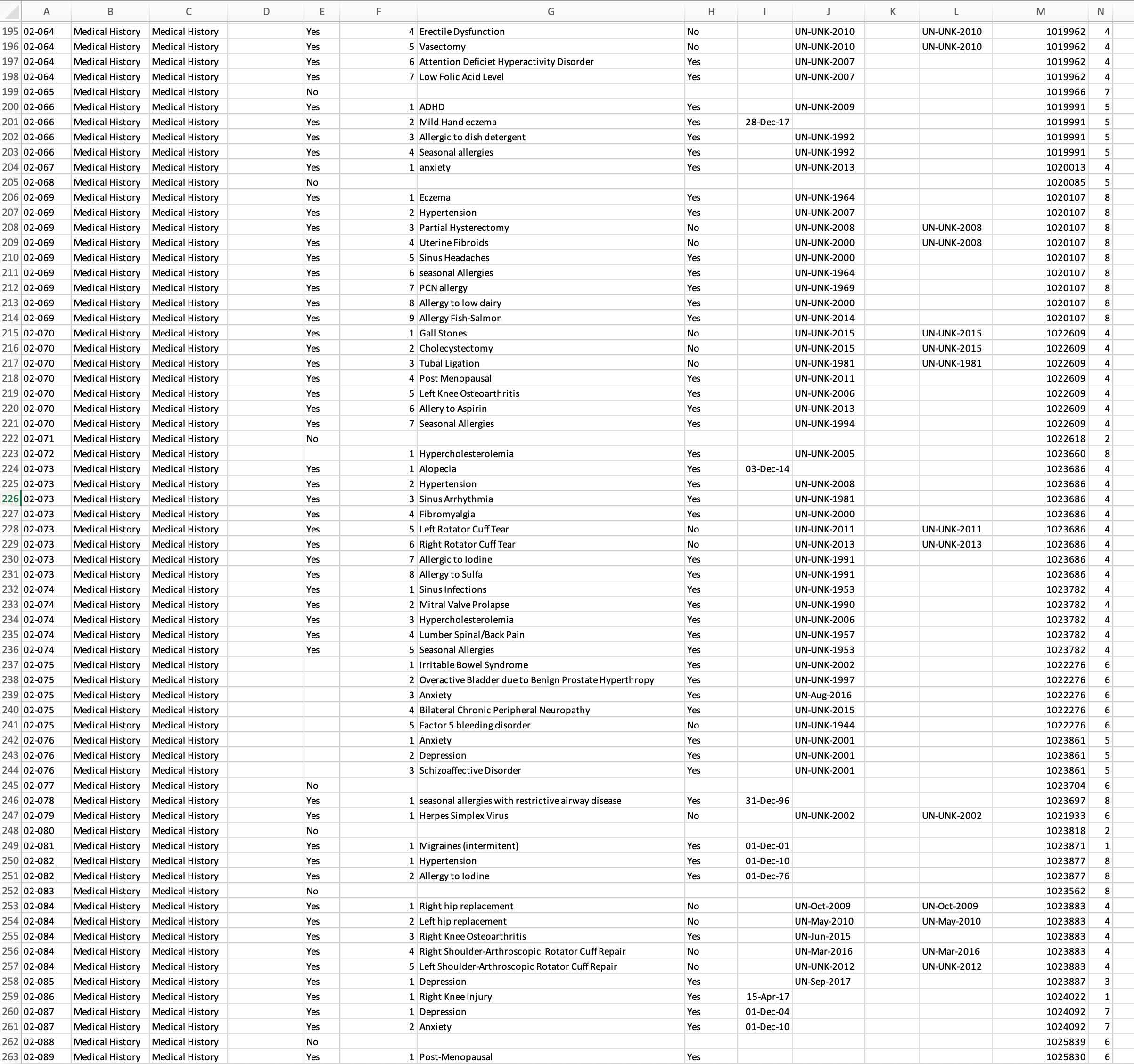
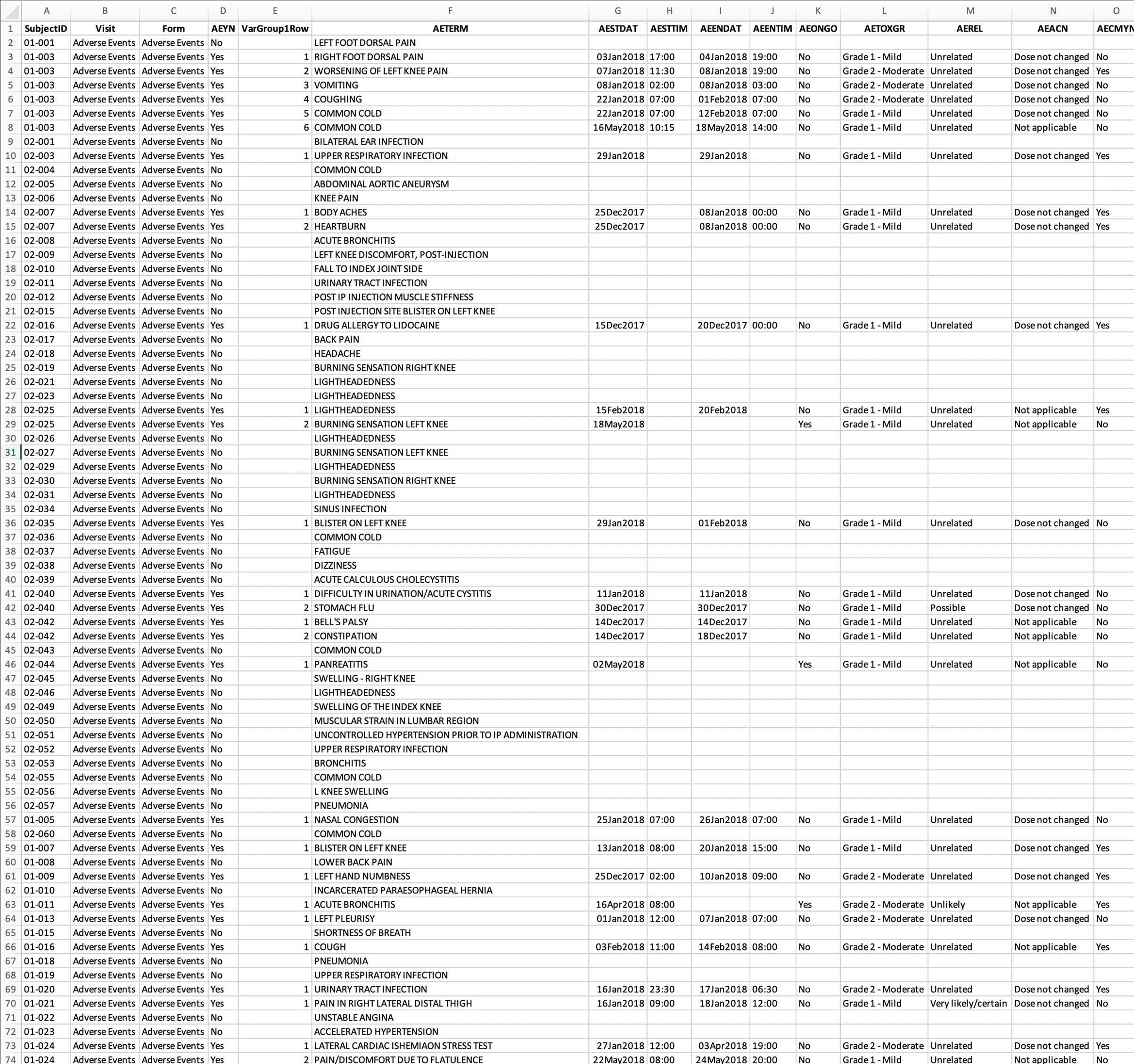
Allow us to integrate typical data sources, such as Adverse Events, Concomitant Medications, and Protocol Deviations, in a way that allows overall safety review and deep dive medical monitor review. A one stop shop. Same data but with customized views for role-based safety and data quality checks.
Consistent, reproducible, and effective: your data in one place. Answers evident. More time for A-C-T-I-O-N! Remove manual integration — step up to our automated process which encourages overall improvement in Patient Safety and Data Integrity.